Abstract
Purpose: We evaluated the tolerability and efficacy of adding lenalidomide to a rituximab and methotrexate combination (R2-MTX) regimen, followed by lenalidomide maintenance as a first-line treatment of patients with primary central nervous system lymphoma (PCNSL).
Patients and methods: This multicenter prospective phase Ib/II study, enrolled immunocompetent patients aged 18-75 years with untreated PCNSL. The phase I study increased lenalidomide dose from 15 to 20 to 25 mg. Patients were treated with rituximab 375 mg/m2 and methotrexate 3.5 g/m2 on day 1 and lenalidomide daily for 14 days every 3-week cycle. After 6 cycles of the R2-MTX regimen, responsive patients received lenalidomide maintenance therapy(lenalidomide 25 mg daily for 21 days every 4-week cycle for 2 years).ASCT was optional in eligible patients before lenalidomide maintenance. The primary phase I endpoint was the maximum tolerated dose of lenalidomide. The primary phase II endpoint was the 2-year progression-free survival (PFS).
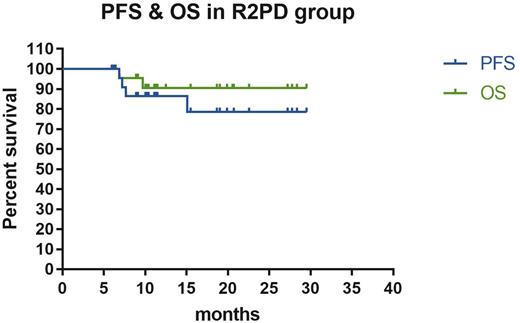
Results: Eleven patients were enrolled in phase Ib of the study. No Dose-limiting toxicities were observed, and the recommend phase II dose of lenalidomide was 25mg. In phase II, 23 patients were treated. The median follow-up period was 14.2 months (range, 8.0-33.4), the estimated 2-year PFS and OS were 61.5%(95% CI 50.7-91.8 ) and 81.7%(95% CI 57.4-92.9), respectively. In the PR2D group, the objective response rate was 92.%, and estimated 2-year PFS and OS were 78.5%(95% CI 50.7-91.8) and 90.4%(95% CI 66.8-97.5). Compared with historical controls in our center, 2-year PFS were significantly improved (P = 0.0088). Hematological toxicities, GI symptoms and infection were the most frequent adverse events, and grade 3/4 toxicities were seen in 62.5% of patients. Quality of life was stabilized during the lenalidomide maintenance phase.
Conclusion: R2-MTX regimen supported the integration of lenalidomide to HD-MTX in induction therapy and maintenance as front-line treatment PCNSL patients with good safety, response rate and 2-year PFS. More importantly, the REMLA trial suggested lenalidomide maintenance could be an option instead of WBRT/ASCT consolidation even in younger patients. Randomized trials that incorporate this regimen are needed to determine its efficacy compared with other strategies.
Disclosures
No relevant conflicts of interest to declare.
OffLabel Disclosure:
Lenalidomide is approved for multiple myeloma treatment. Although it has been used in DLBCL as first-line or salvage treatment, it has never been approved for DLBCL treatment. However, many evidences demonstrated lenalidomide had good response in rrPCNSL. In this trial, we evaluated the efficacy and safety of R2-MTXÃÆ'Æ’&hibar;ÃÆ'ââ,¬Å¡Ãƒâ€šÃ,¼ÃÆ'ââ,¬Â¹ÃƒÂ¢Ã¢â€šâ,¬Ã, lenalidomideÃÆ'Æ’&hibar;ÃÆ'ââ,¬Å¡Ãƒâ€šÃ,¼ÃÆ'ââ,¬&brkbar;ââ‚â,¬Ã¢â€žÂ¢rituximab and MTXÃÆ'Æ’&hibar;ÃÆ'ââ,¬Å¡Ãƒâ€šÃ,¼ÃÆ'Ã,¢Ã¢ââ,¬Å¡Ã¢â€šâ,¬Ãƒâ€šÃ,° in newly-diagnosed PCNSLÃÆ'ƒÂÃ,£ÃÆ'Ã,¢Ã¢â‚â,¬Ã...¡Ã¢ââ,¬Å¡Ã¢â€šâ,¬ÃƒÆ'Ã,¢Ã¢ââ,¬Å¡Ã¢â€šâ,¬Ãƒâ€šÃ,¹
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal